June 21, 2024
5 min read
Cheaper Versions of Popular New Weight-Loss Drugs Are Being Developed
As the patents on various weight-loss drugs near expiration, companies in India and China are vying to make lower-cost versions that will widen access to such treatments
By Smriti Mallapaty & Nature magazine
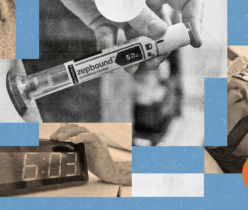
Pharmaceutical companies expect that demand for anti-obesity drugs will increase as price comes down.
Imago/Alamy Stock Photo
Blockbuster weight-loss drugs such as Wegovy could soon become a lot cheaper — and reach more people — thanks to Chinese and Indian pharmaceutical companies. A long queue of companies is developing copies of the complex biological drugs, and some are racing to create modified or improved versions to compete in the global market.
“There is huge potential for companies from India, China, that can help create access to these drugs,” says Abhijit Zutshi, chief commercial officer of the pharmaceutical giant Biocon, headquartered in Bengaluru, India, who oversees its generics business and is based in Woodbridge, New Jersey.
These companies will “bring drugs to patients who need it, at a cost that can be afforded”, says Guy Rutter, a cell biologist at the University of Montreal, Canada.
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Globally, one billion people have obesity or are overweight — and hundreds of millions of them live in India and China. Lei Qian, vice-president of clinical development at Innovent Biologics in Shanghai, China, says that “demand for anti-obesity drugs is very strong” in the country.
In the past few years, a new class of weight-loss drugs has taken the world by storm. They work by mimicking a naturally occurring hormone called glucagon-like peptide 1 (GLP-1). GLP-1 not only regulates blood sugar levels, but also binds to receptors in the brain that control appetite and receptors in the gut that slow digestion and contribute to a feeling of being full.In 2014, the US Food and Drug Administration (FDA) approved the first class of GLP-1 drugs for weight loss. Liraglutide, marketed as Saxenda by Novo Nordisk, based in Bagsværd, Denmark, is a daily injection that can trigger an average body-weight loss of 8% after a year.
The next generation of these drugs required fewer injections and led to greater weight loss. In 2021, the FDA approved semaglutide, sold as Wegovy by Novo Nordisk, as a weight-loss treatment, and in 2023 it approved tirzepatide, marketed as Zepbound by Eli Lilly, based in Indianapolis, Indiana. Tirzepatide mimics GLP-1 as well as another hormone that stimulates insulin secretion, known as glucose-dependent insulinotropic polypeptide (GIP).
Semaglutide and tirzepatide are weekly injections that can induce an average body-weight reduction of between 12% and 18%. In addition to their slimming effects, these drugs are marketed under different brand names, such as Ozempic, as treatments for type 2 diabetes. Clinical trials have also shown that they can reduce the risk of kidney failure, tame inflammation and protect against serious heart problems.
But the drugs are expensive — a month’s worth of semaglutide or tirzepatide injections can cost upwards of US$1,000. One way that Indian and Chinese pharmaceutical companies plan to slash that price is by developing biosimilars, cheaper versions of expensive brand-name drugs. Unlike generics, which are exact copies of chemically synthesized branded drugs, biosimilars very closely resemble their reference product and are derived from modified living organisms, such as yeast.
Companies are preparing to release biosimilar versions of GLP-1 drugs when patent protections lift in different markets. In China, the patent for liraglutide has already expired, and the one for semaglutide will expire in 2026 in India and China.
Companies are also innovating in how they synthesize the drugs, fill the vials and manufacture the injection devices, says Zutshi, whose company is focusing on developing biosimilars for the global market.
How much the cost will drop “is yet to be seen”, says Zutshi. It could be cut in half, or be one-tenth the current price, he says. If drugs are cheaper, more people will use them, thus expanding demand, he adds. This, in turn, could have a “ripple effect” in which companies reduce the price of existing weight-loss drugs “in anticipation of the competition coming”.
No weight-loss biosimilars have yet been approved in India, but Glenmark Pharmaceuticals in Mumbai, India, launched Lirafit, a liraglutide biosimilar, in January for treating type 2 diabetes. Glenmark said in a press release that, at some 100 rupees a day (US$1.20), Lirafit costs 70% less than existing therapies. Biocon has also developed a liraglutide biosimilar, which was approved by the UK drug regulator for type 2 diabetes in March but their product is not on the market yet. Biocon says it is developing semaglutide biosimilars to launch in select countries as soon as 2026.
China’s drug administration has approved two GLP-1 drugs produced by Chinese companies for weight loss. The first is a liraglutide biosimilar marketed as Liluping, made by Huadong Medicine in Hangzhou. The second is beinaglutide, marketed as Feisumei by Benemae Pharmaceutical Corporation in Shanghai.
Several companies in China are ready to pounce on the semaglutide market as soon as the patents expire in 2026. Half a dozen companies have launched late-stage clinical trials of semaglutide injections. One of them, Hangzhou Jiuyuan Gene Engineering, has completed late-stage clinical trials for its semaglutide biosimilar for treatment of type 2 diabetes, and could be the first to enter the Chinese market. A pending court decision on whether the semaglutide patent applies in China could see these drugs enter the market even sooner. Late-stage trials for the company’s semaglutide biosimilar targeting weight loss are expected to start this year.
The arrival of semaglutide biosimilars will be a “game changer” and competition “will be severe”, says Lei.
Beyond biosimilars, companies in India and China are also developing new obesity drugs. For example, Mumbai-based Sun Pharmaceuticals has developed a molecule called GL0034 that has slight tweaks to its chemistry that distinguish it from semaglutide and alter the way it interacts with insulin-secreting cells and nerve cells, says Rutter, who is a consultant for the company. “Not only is the ambition to have a cheaper product, but to have something which is as good, if not better” than semaglutide, he says.
Early-stage clinical trials of GL0034 have been “very encouraging”, says Rajamannar Thennati, executive vice-president for Sun Pharma, who is based in Vadodara, India. The results in three dozen participants show that high doses can lead to body-weight reductions of up to 10% after two months, according to a press release, but Thennati says it could take up to five years for the molecule to hit the market.Sun Pharma is also in the early stages of developing a dual-target drug to compete with tirzepatide. Its structure is sufficiently different to be proprietary, says Rutter.Another improved drug could arrive in the Chinese market even sooner. Innovent has partnered with Eli Lilly to develop a molecule called Mazdutide that mimics both GLP-1 and the glucagon hormone, which stimulates glucose production. Glucagon helps to increase metabolism and enhances fatty-tissue burning. Results from late-stage clinical trials for Mazdutide will be announced at the American Diabetes Association meeting in Orlando, Florida, next week. Lei says they are “positive”. Preliminary data suggest some trial participants taking high doses lost some 19% of their body weight after close to a year. Lei expects that Mazdutide will be approved for weight loss by China’s drug regulator in the first half of 2025, which would make it the first of this class of dual-target drugs to be approved globally.
This article is reproduced with permission and was first published on June 19, 2024.
Smriti Mallapaty is a senior reporter at Nature.
First published in 1869, Nature is the world’s leading multidisciplinary science journal. Nature publishes the finest peer-reviewed research that drives ground-breaking discovery, and is read by thought-leaders and decision-makers around the world.
Learn and share the most exciting discoveries, innovations and ideas shaping our world today.
Follow Us:
Scientific American is part of Springer Nature, which owns or has commercial relations with thousands of scientific publications (many of them can be found at www.springernature.com/us). Scientific American maintains a strict policy of editorial independence in reporting developments in science to our readers.
© 2024 SCIENTIFIC AMERICAN, A DIVISION OF SPRINGER NATURE AMERICA, INC.
ALL RIGHTS RESERVED.