Learn how we are healing patients through science & compassion
‘Smart speaker’ shows potential for better self-management of Type 2 diabetes
Learn how we are fueling innovation
Students from far and near begin medical studies at Stanford
Learn how we empower tomorrow’s leaders
Support teaching, research, and patient care.
Support Lucile Packard Children’s Hospital Stanford and child and maternal health
Stanford School of Medicine
Stanford Health Care
Stanford Children’s Health
Administration
Aging
Big Data
Biochemistry
Bioengineering
Cancer
Cardiovascular Health
Chemical Biology
Chronic Disease
COVID-19
Developmental Biology
Education
Genetics
Global Health
Health Policy
Hearing
Imaging
Immunology
Infectious Disease
Mental Health
Neuroscience
Nutrition
Obituaries
Pain
Patient Care
Pediatrics
Precision Health
Preventive Medicine
Sleep
Stem Cells
Surgery
Technology
Transplantation
Urology
Women’s Health
A Stanford Medicine study found that metformin, a commonly prescribed diabetes drug associated with moderate weight loss, stimulates production of lac-phe, a molecule abundant after exercise.
March 18, 2024 – By Krista Conger
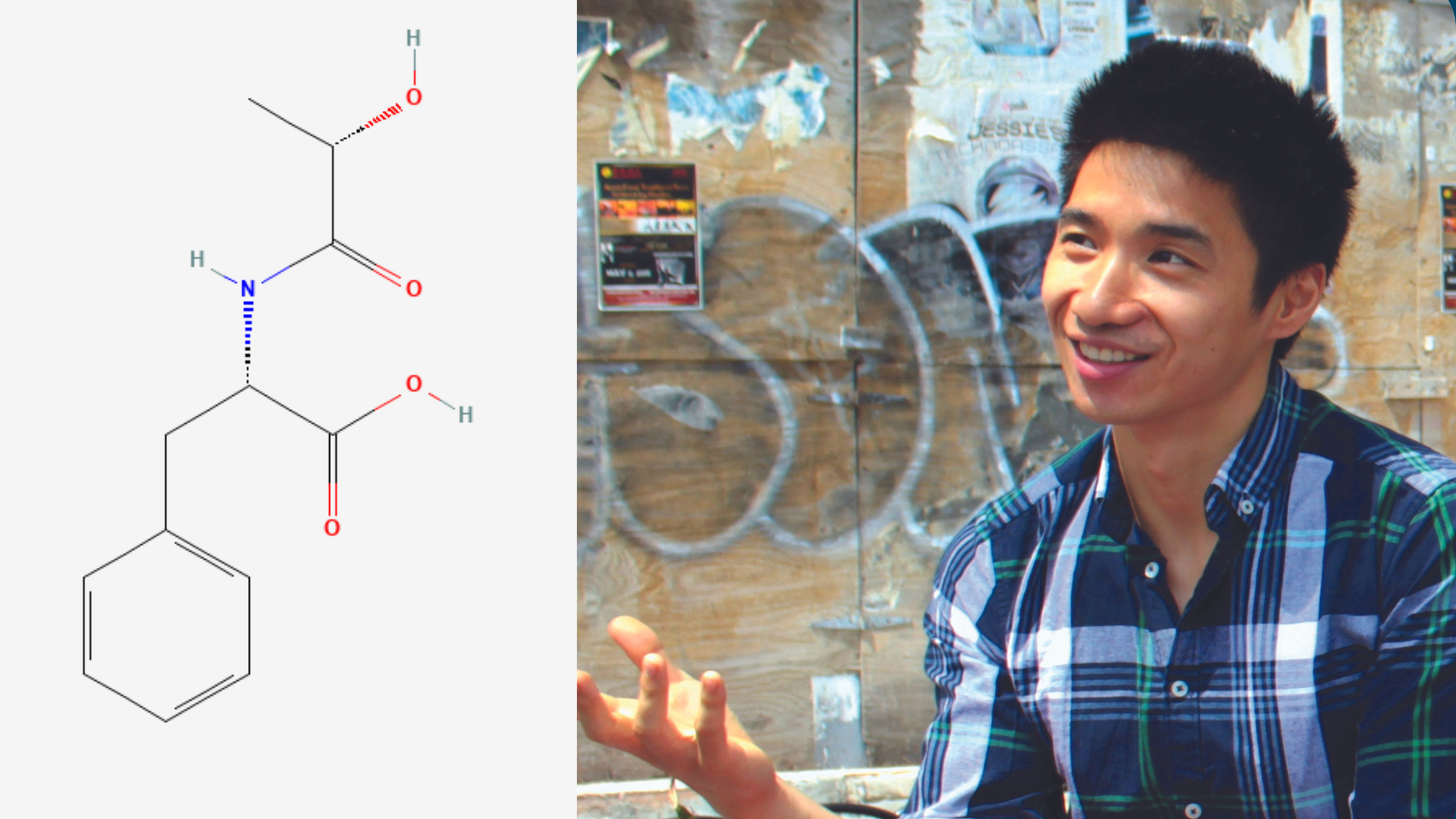
Jonathan Long co-discovered lac-phe, known as the "anti-hunger" molecule.
Courtesy Jonathan Long
An “anti-hunger” molecule produced after vigorous exercise is responsible for the moderate weight loss caused by the diabetes medication metformin, according to a new study in mice and humans. The molecule, lac-phe, was discovered by Stanford Medicine researchers in 2022.
The finding, made jointly by researchers at Stanford Medicine and at Harvard Medical School, further cements the critical role the molecule, called lac-phe, plays in metabolism, exercise and appetite. It may pave the way to a new class of weight loss drugs.
“Until now, the way metformin, which is prescribed to control blood sugar levels, also brings about weight loss has been unclear,” said Jonathan Long, PhD, an assistant professor of pathology. “Now we know that it is acting through the same pathway as vigorous exercise to reduce hunger. Understanding how these pathways are controlled may lead to viable strategies to lower body mass and improve health in millions of people.”
Long, who is supported by the Knight Initiative for Brain Resilience, and Mark Benson, MD, PhD, an assistant professor of medicine at Harvard Medical School, are co-senior authors of the study, which was published on March 18 in Nature Metabolism. Postdoctoral scholar Shuke Xiao, PhD, is the lead author of the study.
Many people with diabetes who are prescribed metformin lose around 2% to 3% of their body weight within the first year of starting the drug. Although this amount of weight loss is modest when compared with the 15% or more often seen by people taking semaglutide drugs such as Ozempic and Wegovy, the discoveries that led to those drugs also grew from observations of relatively minor, but reproducible, weight loss in people taking first-generation versions of the medications.
When Long and colleagues at Baylor University discovered lac-phe in 2022, they were on the hunt for small molecules responsible for curtailing hunger after vigorous exercise. What they found was a Frankenbaby of lactate — a byproduct of muscle fatigue — and an amino acid called phenylalanine. They dubbed the hybrid molecule lac-phe and went on to show that it’s not only more abundant after exercise but it also causes people (as well as mice and even racehorses) to feel less hungry immediately after a hard workout.
“There is an intimate connection between lac-phe production and lactate generation,” Long said. “Once we understood this relationship, we started to think about other aspects of lactate metabolism.”
Metformin was an obvious candidate because as it stimulates the breakdown of glucose (thus reducing blood sugar levels) it can trigger the generation of lactate.
The researchers found that obese laboratory mice given metformin had increased levels of lac-phe in their blood. They ate less than their peers and lost about 2 grams of body weight during the nine-day experiment.
Long and his colleagues also analyzed stored blood plasma samples from people with Type 2 diabetes before and 12 weeks after they had begun taking metformin to control their blood sugar. They saw significant increases in the levels of lac-phe in people after metformin compared with their levels before treatment. Finally, 79 participants in a large, multi-ethnic study of atherosclerosis who were also taking metformin had significantly higher levels of lac-phe circulating in their blood than those who were not taking the drug.
“It was nice to confirm our hunch experimentally,” Long said. “The magnitude of effect of metformin on lac-phe production in mice was as great as or greater than what we previously observed with exercise. If you give a mouse metformin at levels comparable to what we prescribe for humans, their lac-phe levels go through the roof and stay high for many hours.”
Further research revealed that lac-phe is made by intestinal epithelial cells in the animals; blocking the ability of mice to make lac-phe erased the appetite suppression and weight loss previously observed.
Finally, a statistical analysis of the people in the atherosclerosis study who lost weight during the several-year study and follow-up period found a meaningful association between metformin use, lac-phe production and weight loss.
“The fact that metformin and sprint exercise affect your body weight through the same pathway is both weird and interesting,” Long said. “And the involvement of the intestinal epithelial cells suggests a layer of gut-to-brain communication that deserves further exploration. Are there other signals involved?”
Long noted that, while semaglutide drugs are injected into the bloodstream, metformin is an oral drug that is already prescribed to millions of people. “These findings suggest there may be a way to optimize oral medications to affect these hunger and energy balance pathways to control body weight, cholesterol and blood pressure. I think what we’re seeing now is just the beginning of new types of weight loss drugs.”
Researchers from Beth Israel Deaconess Medical Center, Harbor-UCLA Medical Center, Cedars-Sinai Medical Center, Baylor College of Medicine, the University of Colorado, the University of Virginia and the Broad Institute contributed to the work.
The study was funded by the National Institutes of Health (grants GM113854, K08HL145095, DK124265, DK136526, HHSN2682015000031, HSN26800004, UM1DK078616 and 1R01HL151855), a Stanford School of Medicine’s Dean’s Fellowship, the American Heart Association, Wu Tsai Human Performance Alliance, Knight Initiative for Brain Resilience and Stanford Diabetes Research Center.
About Stanford Medicine
Stanford Medicine is an integrated academic health system comprising the Stanford School of Medicine and adult and pediatric health care delivery systems. Together, they harness the full potential of biomedicine through collaborative research, education and clinical care for patients. For more information, please visit med.stanford.edu.
Stanford Medicine researchers and their collaborators have identified a molecule that staves off hunger post-exercise.
Psychiatry’s new frontiers
Contact Us
For Media
For Faculty & Staff
News Center
Scope
Stanford Medicine Magazine
Stanford Medicine Children’s Health Blog
About
Contact
Maps & Directions
Careers
Basic Science Departments
Clinical Science Departments
Academic Programs
Vision